Hot Topics
Vol. 59, No. 10, October(2010)
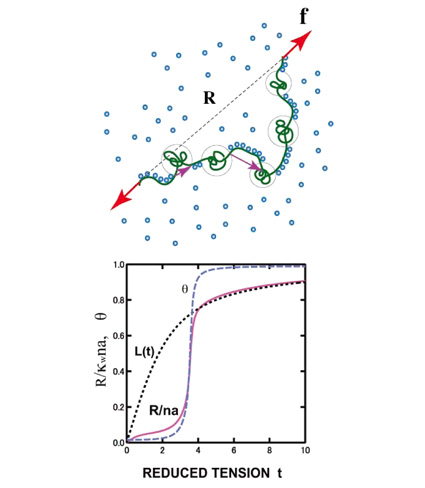
| Cooperative Hydration of Poly(N-isopropylacrylamide) Fumihiko TANAKA Department of Polymer Chemistry, Kyoto University |
||
|
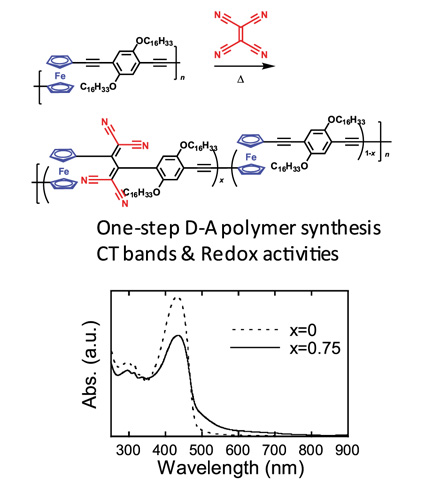
| Postfunctionalization of Conjugated Polymers by Alkyne-Acceptor Click Chemistry Tsuyoshi MICHINOBU Global Edge Institute, Tokyo Institute of Technology, and PRESTO, JST |
||
|
| Organic Polymer-Assisted Formation of Microspheres Consisting of Pristine Fullerenes Masayuki TAKEUCHI Organic Nanomaterials Center, National Institute for Materials Science |
||
|
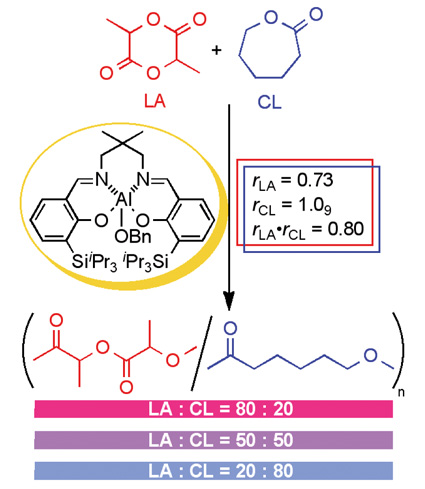
| Ideal Copolymerization of Lactide with e-Caprolactone Azusa AKITA, Ryohei ISHII, and Nobuyoshi NOMURA Lab. Polym. Chem., Grad. Sch. Bioagr. Sci., Nagoya University |
||
|
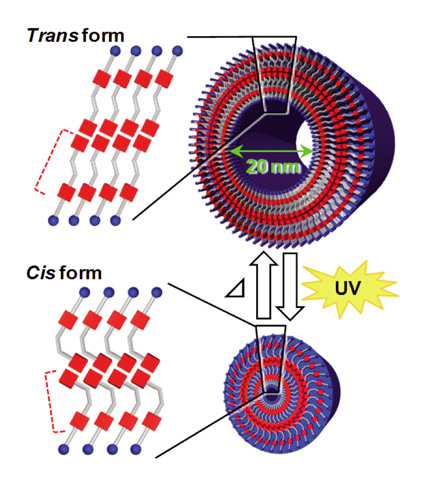
| Construction of Photo-Responsive Organic Nanotubes Aiming at Controllable Guest Release Naohiro KAMETA, Asuka TANAKA, Haruhisa AKIYAMA, Hiroyuki MINAMIKAWA, Mitsutoshi MASUDA, and Toshimi SHIMIZU Nanotube Research Center, National Institute of Advanced Industrial Science and Technology (AIST) |
||
|
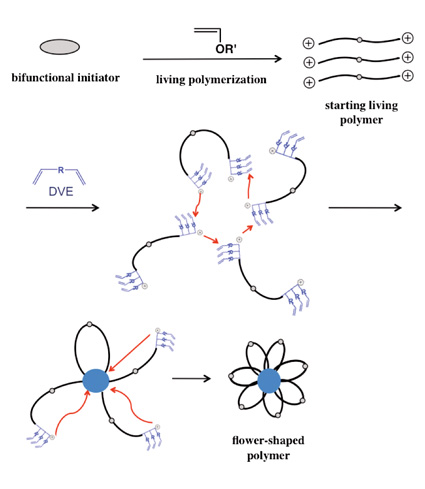
| Selective Synthesis of Flower-Shaped Polymers by Living Cationic Polymerization Shokyoku KANAOKA, Aya YANAGISAWA, Naoto YAGI, and Sadahito AOSHIMA Graduate School of Science, Osaka University |
||
|
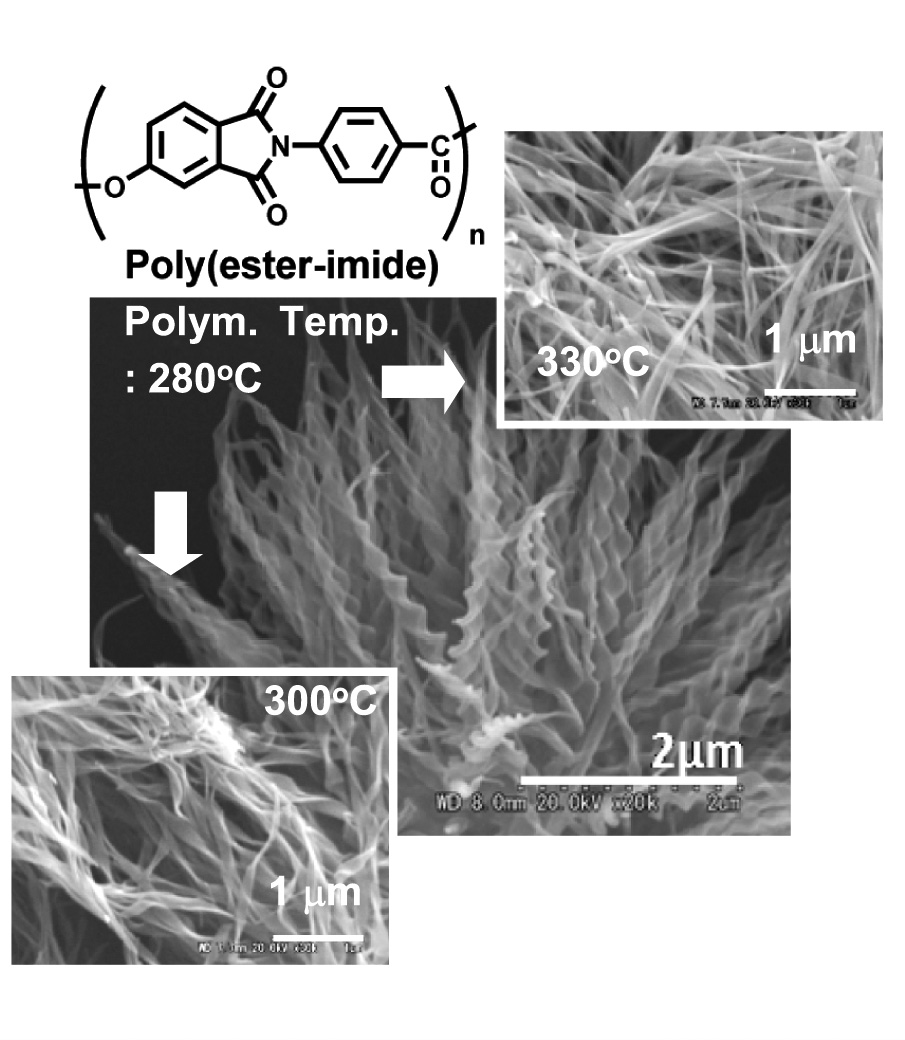
| Helical Crystals of Poly (ester imide) Miki NAKAGAWA1, Kanji WAKABAYASHI1, Yuko MAKI1, Tetsuya UCHIDA2, Shinichi YAMAZAKI1, and Kunio KIMURA1 1Graduate School of Environmental Science, Okayama University 2Graduate School of Natural Science and Technology, Okayama University |
||
|
| Planar Chiral [2.2]Paracyclophanes and Their Conjugated Polymers Yasuhiro MORISAKI, Lin LIN, and Yoshiki CHUJO Graduate School of Engineering, Kyoto University |
||
|
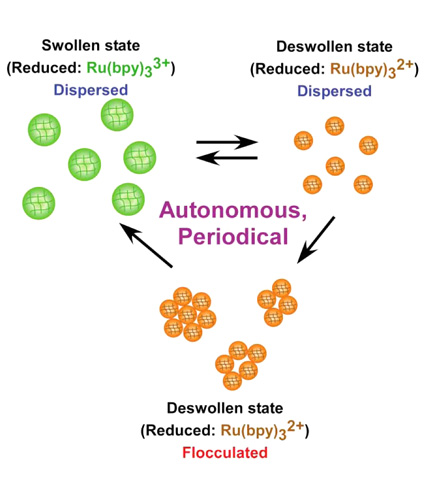
| Control of Temporal Structures for Polymer Particles Daisuke SUZUKI* and Ryo YOSHIDA International Young Researchers Empowerment Center, Shinshu University |
||
|